 
In such a case, the interaction between these molecules also affects the polarity of the molecule, and often, a non-polar molecule may also become polar. Things change when a molecule is surrounded by similar or dissimilar molecules. In the above explanation of the dipole moment and polarity of a molecule, it is assumed that the molecule is isolated. One should remember that the dipole moment of a bond is never negative. unit of dipole moment is Cm (Coulomb meters) or D (Debye).ġ Debye = when 3.336 × 10−30 Coulomb of charge is separated by 1 meter. R = distance between the atoms involved in the bond Here, q = the absolute charge separation between the atoms involved in the bond Mathematically, the dipole moment of a bond is expressed as: The polarity of a bond can be quantitatively expressed in the terms of dipole moment. Thus, angle(s) between dipoles also affect the net dipole moment between different dipoles. Different dipoles can be added or subtracted following the mathematics of vectors. The polarity of a molecule can be determined by checking the polarity of each of the bonds in the molecule, and the arrangement of these bonds in space.Īlso, the electronegativity of each atom also plays a major role in affecting the polarity of the molecule. This summary may seem a little confusing, but going through the following article will make it more understandable. Due to this, the individual Se-F bond’s polarity is not canceled and thus SeF4 has a net dipole moment. 
This molecule has a see-saw shape, with Selenium as the central atom and Fluorine atoms surrounding it in a see-saw fashion in accordance with VSEPR theory. The arrangement of four F atoms and a lone pair in SeF4 is not symmetrical (we will see why it is asymmetrical in later discussion).į atom is more electronegative than Se atom, thus the shared pairs of electrons within the Se-F bond are pulled more towards the F atom- giving rise to a dipole with its head at F atom and tail at Se atom. it has trigonal bipyramidal geometry and a see-saw shape. The question is, why SeF4 is polar in nature. Let us study the concept of polarity in detail. The lone pair and arrangement of four fluorine atoms around selenium cause the unequal distribution of charge across the molecule. Fluorine is more electronegative than Selenium due to which the Se-F bond is also polar. The asymmetric shape of the molecule results in the non-zero value of dipole moment. So, is SeF4 polar or nonpolar? SeF4 is a polar molecule because of its trigonal bipyramidal geometry and see-saw shape. In this article, we will explore whether selenium tetrafluoride is polar and for that we will take the help of some well-accepted theories in chemistry. SeF4 has many important applications in organic synthesis reactions. SeF4 is commercially produced by reacting selenium with chlorine trifluoride (ClF3). For this reason, SeF4 is rarely used for this purpose. SeF4 quickly reacts with water molecules to get hydrolyzed and releases hydrogen fluoride, which is a very harmful substance. It is a more efficient fluorinating reagent than SF4, as SeF4 is a liquid and could be handled easily under mild conditions. It is used as a very efficient selective fluorinating agent and has been used to fluorinate aldehydes, ketones, alcohols and carboxylic acids, activated carbon surfaces. Se has a +4 oxidation state in this molecule. 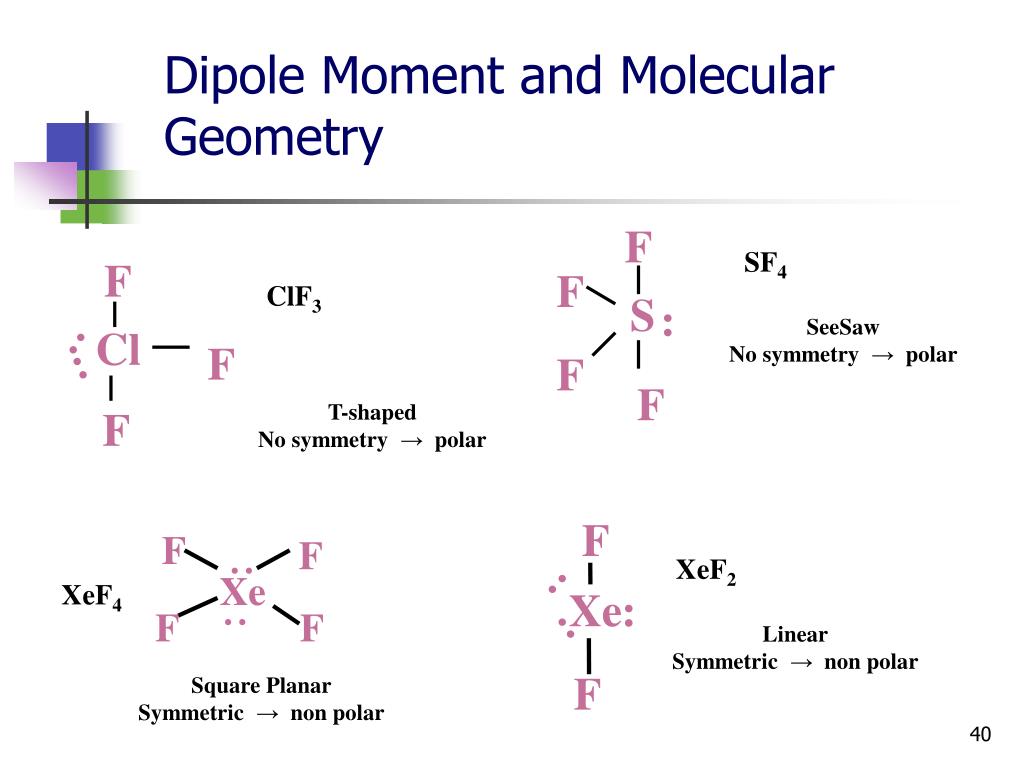
SeF4, selenium tetrafluoride, is a colorless toxic and poisonous liquid that boils at a temperature close to that of water. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
